Understanding Peptide Purity: HPLC and Mass Spectrometry Analysis
Discover how HPLC and mass spectrometry ensure the highest quality research peptides and what purity percentages mean for your research.
Understanding Peptide Purity: HPLC and Mass Spectrometry Analysis
When purchasing research peptides, purity is one of the most critical quality indicators. At Protopep, all our peptides are analyzed using High-Performance Liquid Chromatography (HPLC) and Mass Spectrometry (MS) to guarantee ≥99% purity.
What is Peptide Purity?
Peptide purity refers to the percentage of the desired peptide sequence present in the product, excluding:
- Deletion sequences (missing amino acids)
- Truncated sequences (incomplete chains)
- Modified sequences (unintended modifications)
- Salts and counterions
- Residual solvents
HPLC Analysis Explained
How HPLC Works
High-Performance Liquid Chromatography separates peptide components based on their chemical properties:
- Sample is dissolved in a mobile phase (solvent)
- Solution passes through a stationary phase (column)
- Different components separate based on their interaction with both phases
- Detector measures each component as it exits the column
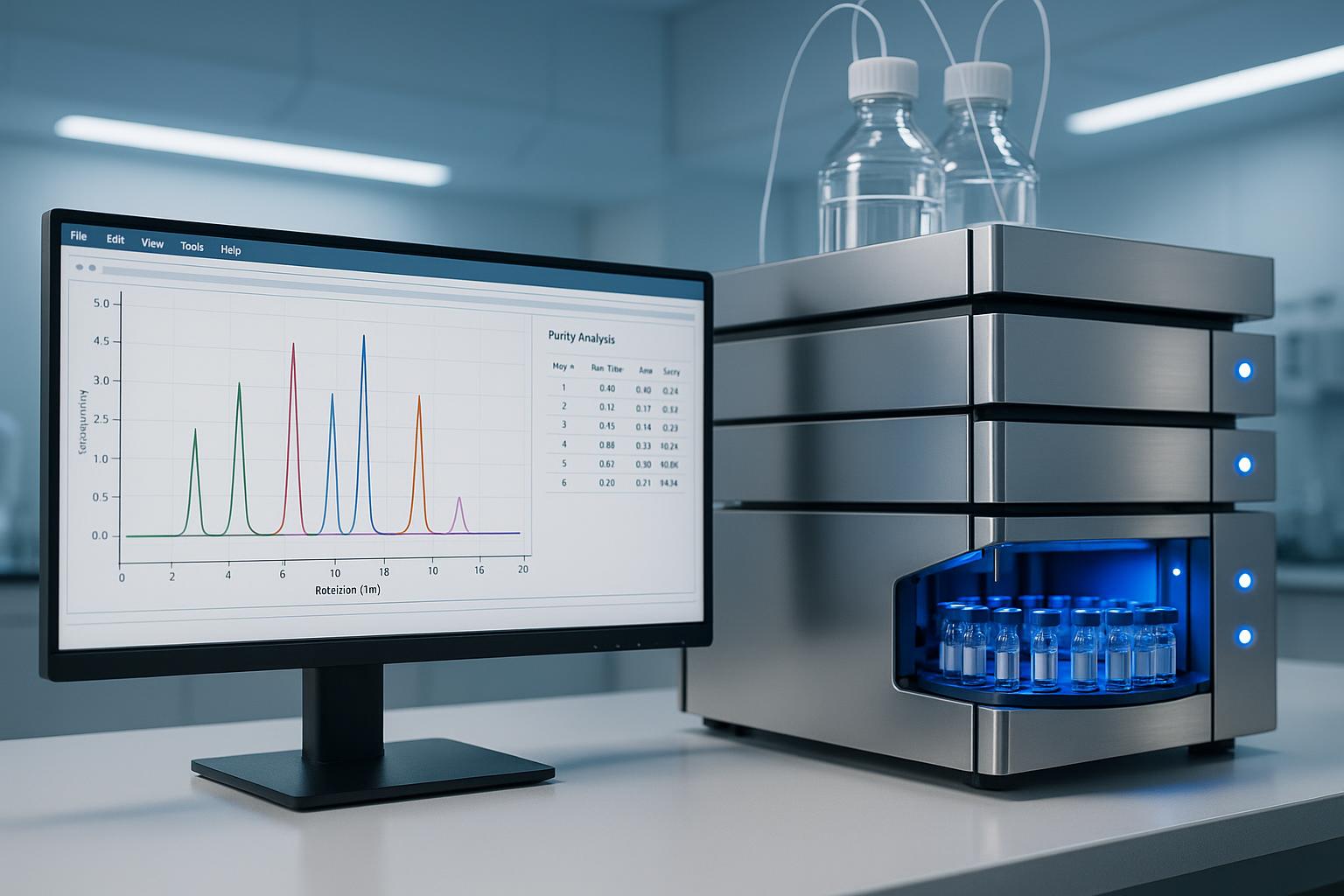
- Results displayed as a chromatogram showing peaks
Reading HPLC Results
- Main peak represents the target peptide
- Peak area indicates relative quantity
- Smaller peaks represent impurities
- Purity calculated as: (Target Peak Area / Total Peak Area) × 100
Mass Spectrometry Analysis
Purpose of MS Analysis
While HPLC measures purity, Mass Spectrometry confirms identity:
- Verifies correct molecular weight
- Confirms peptide sequence
- Detects modifications or errors
- Provides additional purity information
MS Techniques Used
- ESI-MS (Electrospray Ionization): Most common for peptides
- MALDI-TOF: Used for larger peptides
- LC-MS: Combined liquid chromatography and mass spec
Why Purity Matters in Research
Impact on Results
Higher purity peptides provide:
- More consistent results
- Better reproducibility
- Accurate dose-response relationships
- Reduced confounding variables
- Enhanced safety profiles
Purity Grades
- ≥95%: Suitable for most research applications
- ≥98%: Recommended for sensitive assays
- ≥99%: Required for clinical and pharmaceutical research (Protopep standard)
Certificate of Analysis (CoA)
Every Protopep peptide includes a CoA containing:
- HPLC chromatogram
- Mass spectrometry data
- Calculated purity percentage
- Molecular weight (expected vs. observed)
- Batch number and manufacturing date
- Storage recommendations
Quality Control at Protopep
Our Testing Process
- Synthesis using solid-phase peptide synthesis (SPPS)
- Initial HPLC purification
- HPLC analysis for purity determination
- Mass spectrometry for identity confirmation
- Lyophilization (freeze-drying)
- Final quality check and CoA generation
- Third-party verification for select batches
Why We Guarantee ≥99% Purity
Research-grade peptides require the highest quality standards:
- Eliminates variables from impurities
- Ensures reproducible results
- Meets international research standards
- Provides confidence in your experimental data
Common Impurities in Lower-Grade Peptides
Synthesis-Related Impurities
- Deletion sequences: Missing amino acids
- Truncated peptides: Incomplete chains
- Diastereomers: Wrong stereochemistry
- Acetylated/TFA-capped peptides: Incomplete deprotection
Purification-Related Issues
- Residual TFA (trifluoroacetic acid)
- Salts and counterions
- Residual solvents
- Aggregates
Interpreting Your CoA
Key Information to Check
- Purity percentage (should be ≥99%)
- Molecular weight match (observed vs. expected)
- HPLC chromatogram (single dominant peak)
- Batch number (for traceability)
- Expiration date
- Storage conditions
Red Flags
Be cautious if:
- Purity is not clearly stated
- No HPLC chromatogram provided
- Mass spec data is missing
- Multiple large peaks in HPLC
- Molecular weight doesn't match
Conclusion
Understanding peptide purity and analytical methods helps you make informed decisions about research peptide quality. At Protopep, our commitment to ≥99% purity, verified by HPLC and mass spectrometry, ensures you receive the highest quality peptides for your research applications.
Always request and review the Certificate of Analysis for any research peptide purchase to verify quality and authenticity.
Tags
Stay Updated with Latest Research
Get exclusive access to new case studies, research insights, and peptide therapy protocols delivered straight to your inbox.
No spam. Unsubscribe anytime. We respect your privacy.